Understanding Glioblastoma
Glioblastoma (GBM), formerly known as Glioblastoma Multiforme, represents the most aggressive form of primary brain cancer. This comprehensive guide provides essential information about diagnosis, treatment options, and the latest advances in immunotherapy, specifically designed for patients, families, and healthcare professionals seeking expert knowledge.
Moreover, understanding this complex condition empowers patients to make informed decisions about their care. Consequently, we have compiled the most current scientific evidence and clinical expertise to help you navigate this challenging journey.
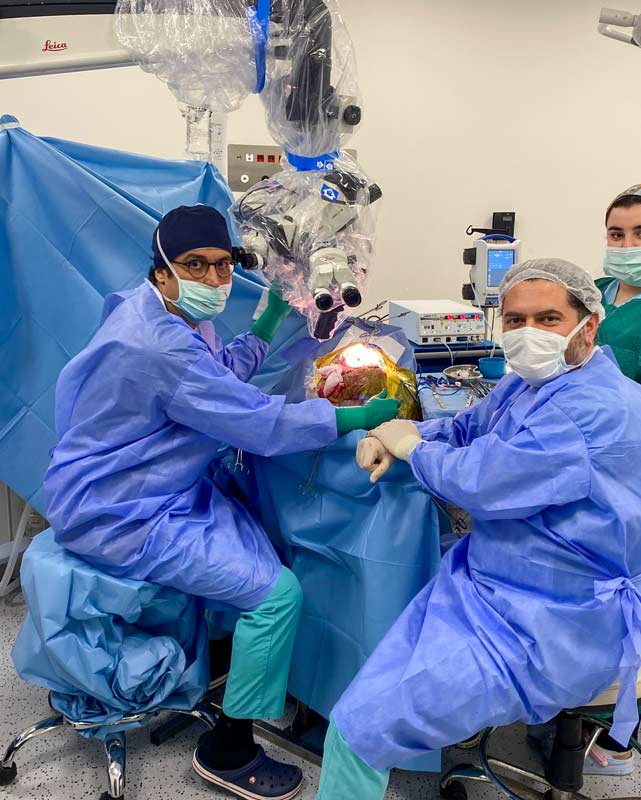
Intraoperative view during GBM microsurgical resection
Table of Contents
What is Glioblastoma?
Glioblastoma (GBM), formerly known as Glioblastoma Multiforme, stands as the most prevalent and aggressive primary brain tumor affecting adults worldwide. The term "Glioblastoma Multiforme" was historically used due to the tumor's heterogeneous appearance, but current WHO classification now uses the simplified name "Glioblastoma." It represents approximately 47.7% of all malignant central nervous system tumors, making it a significant focus of neurosurgical and oncological research.
Definition and Classification
Glioblastoma (GBM), formerly referred to as Glioblastoma Multiforme and also known as a Grade IV astrocytoma, originates from the brain's supportive glial cells called astrocytes. The WHO 2021 classification officially adopted the simplified name "Glioblastoma, IDH-wildtype" while the historical term "Glioblastoma Multiforme" remains widely recognized in medical literature. Furthermore, the World Health Organization classifies this tumor as the highest grade (Grade IV) due to its highly aggressive nature and rapid growth pattern.
Importantly, GBM demonstrates several distinguishing characteristics that set it apart from lower-grade brain tumors. First, it exhibits rapid cellular proliferation. Second, it shows extensive infiltration into surrounding healthy brain tissue. Third, it develops areas of necrosis (dead tissue) within the tumor mass. Finally, it promotes abnormal blood vessel formation (neovascularization).
Key Point
GBM can arise de novo (primary GBM) without any precursor lesion, or it can evolve from lower-grade astrocytomas (secondary GBM). Primary GBM accounts for approximately 90% of cases and typically occurs in older patients.
Why is GBM So Challenging?
GBM presents unique treatment challenges that make it particularly difficult to manage. Specifically, several factors contribute to its complexity:
- Strategic Location: The tumor resides within the brain, limiting aggressive treatment options
- Inherent Resistance: GBM cells demonstrate natural resistance to conventional therapy
- Limited Brain Repair: The brain has minimal capacity to repair damage caused by both the tumor and treatment
- Infiltrative Growth: Malignant cells migrate into adjacent healthy brain tissue, making complete removal impossible
- Blood-Brain Barrier: This protective barrier limits the effectiveness of many drugs
- Tumor Heterogeneity: Different areas of the tumor have different genetic characteristics
Pre-operative and post-operative MRI comparison showing GBM before and after surgical resection
GBM at a Glance
| WHO Grade | Grade IV (Highest) |
| Cell of Origin | Astrocytes (Glial Cells) |
| Peak Age | 55-65 years |
| Common Location | Cerebral Hemispheres (95%) |
| Growth Pattern | Diffusely Infiltrative |
| Metastasis | Rare (Local invasion) |
Types of Brain Gliomas
Understanding the classification of brain tumors helps patients and families comprehend where GBM fits within the spectrum of central nervous system tumors. Consequently, this knowledge aids in understanding treatment decisions and prognosis.
WHO Grading System
The World Health Organization classifies gliomas into four grades based on their histopathological features and biological aggressiveness. Notably, higher grades indicate more aggressive tumor behavior.
Grade I - Pilocytic Astrocytoma
Slow-growing, well-circumscribed tumors that rarely transform into higher-grade lesions. Furthermore, surgical resection often achieves cure.
Grade II - Diffuse Astrocytoma
Slow-growing but infiltrative tumors that can progress to higher grades over time. Additionally, they require careful monitoring and treatment planning.
Grade III - Anaplastic Astrocytoma
Anaplastic astrocytoma is a malignant WHO Grade III brain tumor that represents a critical intermediate stage between low-grade gliomas and glioblastoma. These tumors show increased cellular proliferation, nuclear atypia, and mitotic activity compared to Grade II tumors.
Importantly, anaplastic astrocytomas carry a significant risk of progression to Grade IV glioblastoma, which occurs in approximately 70% of cases. Treatment typically involves surgical resection followed by radiation therapy and chemotherapy. IDH mutation status is a key prognostic factor — patients with IDH-mutant anaplastic astrocytoma generally have better outcomes, with median survival of 3-5 years compared to 1-2 years for IDH-wildtype tumors. Early detection and monitoring of Grade III tumors is essential to prevent malignant transformation to glioblastoma.
Grade IV - Glioblastoma (formerly Glioblastoma Multiforme)
The most aggressive form, characterized by necrosis, microvascular proliferation, and rapid growth. Consequently, it requires immediate and aggressive multimodal treatment.
Primary vs. Secondary GBM
GBM can be classified based on its development pattern, which has important implications for molecular characteristics and patient demographics.
Primary GBM (De Novo)
- Accounts for approximately 90% of all GBM cases
- Arises without evidence of a precursor lesion
- Typically affects older patients (median age 62 years)
- Characterized by EGFR amplification and PTEN loss
- Usually IDH-wildtype status
Secondary GBM
- Accounts for approximately 10% of GBM cases
- Evolves from lower-grade astrocytomas
- Tends to occur in younger patients (median age 45 years)
- Characterized by IDH1/2 mutations and TP53 mutations
- Generally associated with better prognosis
Clinical Significance
Distinguishing between primary and secondary GBM is crucial because secondary GBM patients often have better treatment responses and longer survival times due to their molecular profile.
Epidemiology of Glioblastoma
Understanding the epidemiology of GBM provides valuable context about who is affected by this disease and helps identify populations at higher risk. Furthermore, this data guides research priorities and resource allocation.
Per 100,000
Annual incidence rate in the general population
Years
Median age at diagnosis
Male to Female
Gender ratio showing male predominance
Of Adult Gliomas
GBM's share of all adult brain tumors
Incidence and Demographics
GBM accounts for approximately 47.7% of all malignant brain and central nervous system tumors. Additionally, it represents the most common primary malignant brain tumor in adults, with an incidence of 3.21 per 100,000 population annually.
The median age at diagnosis is 64 years, and the tumor occurs more frequently in men compared to women, with a ratio of approximately 1.6:1. Furthermore, incidence rates vary by ethnicity, with higher rates observed in Caucasian populations compared to other ethnic groups.
Importantly, while GBM predominantly affects adults, it can occur at any age. In children, GBM is less common but tends to behave differently, with distinct molecular characteristics and treatment considerations.
Geographic Variations
Interestingly, the incidence of GBM varies geographically. Western countries report higher incidence rates compared to less developed nations. However, this disparity may reflect differences in diagnostic capabilities, access to healthcare, and reporting practices rather than true biological differences.
Survival Statistics
Unfortunately, the prognosis for GBM remains poor despite advances in treatment. The current statistics underscore the urgent need for continued research and innovative therapies.
Survival Rates
Important Note
These statistics represent averages and do not predict individual outcomes. Many factors influence survival, including age, tumor location, extent of resection, molecular markers, and overall health status.
Risk Factors for Glioblastoma
Understanding the risk factors for GBM helps identify vulnerable populations and guides prevention strategies. However, it is important to note that most GBM cases occur without any identifiable cause.
Ionizing Radiation
Prior exposure to high-dose therapeutic radiation represents the only confirmed environmental risk factor for GBM. Consequently, patients who received cranial radiation for previous conditions face elevated risk.
Genetic Syndromes
Certain hereditary conditions significantly increase GBM risk, including Li-Fraumeni syndrome, neurofibromatosis type 1, Lynch syndrome, and tuberous sclerosis. Nevertheless, these account for only 5-10% of cases.
Demographics
GBM occurs more frequently in males and primarily affects adults between 45-75 years. Additionally, Caucasian populations show higher incidence rates compared to other ethnic groups.
What Does NOT Cause GBM?
Extensive research has found no conclusive association between GBM and: cell phone use, electromagnetic fields, smoking, dietary factors, severe head injury, or common pesticide exposure. Therefore, patients should not blame themselves for developing this condition.
Signs and Symptoms of GBM
GBM symptoms develop through three main mechanisms: direct tissue destruction, increased intracranial pressure, and seizure activity. Importantly, symptoms vary based on tumor location, size, and growth rate.
Common Symptoms
Persistent headaches that worsen over time, often more severe in the morning. These result from increased intracranial pressure caused by tumor growth.
New-onset seizures in adults should always prompt brain imaging. Seizures may be focal, complex partial, or generalized.
Memory problems, difficulty concentrating, and confusion may develop gradually as the tumor affects brain function.
Weakness on one side, speech difficulties, or vision problems depend on tumor location within the brain.
Frontal lobe tumors particularly cause mood swings, personality alterations, and behavioral changes.
Symptoms by Location
| Frontal Lobe | Personality changes, weakness, speech problems |
| Temporal Lobe | Memory issues, hearing problems, seizures |
| Parietal Lobe | Sensory loss, spatial awareness problems |
| Occipital Lobe | Visual field defects, vision loss |
| Motor Cortex | Contralateral weakness or paralysis |
When to Seek Immediate Care
Seek emergency medical attention for: sudden severe headache, new seizures, sudden vision loss, sudden weakness, or rapid deterioration in consciousness.
How is GBM Diagnosed?
Accurate diagnosis of GBM requires sophisticated imaging techniques combined with tissue analysis. Furthermore, molecular profiling has become essential for treatment planning and prognostication.
Advanced Imaging Techniques
Magnetic Resonance Imaging (MRI) serves as the gold standard for GBM diagnosis. Typically, GBM appears as a heterogeneous mass with ring-like contrast enhancement surrounding a necrotic core, accompanied by significant surrounding edema.
Imaging Modalities
- Conventional MRI with Contrast: Primary diagnostic tool showing tumor extent
- MR Spectroscopy (MRS): Analyzes chemical composition of tumor tissue
- Functional MRI (fMRI): Maps critical brain functions for surgical planning
- Diffusion Tensor Imaging (DTI): Visualizes white matter tracts
- Perfusion MRI: Assesses tumor blood flow and vascularity
- PET Imaging: Evaluates metabolic activity and distinguishes recurrence from treatment effects
Tissue Diagnosis & Molecular Analysis
Definitive diagnosis requires tissue obtained through surgical resection or biopsy. Subsequently, neuropathologists examine the tissue for characteristic features and perform molecular testing.
Key Molecular Markers
| IDH1/IDH2 | Mutation status differentiates primary vs secondary GBM |
| MGMT | Promoter methylation predicts temozolomide response |
| EGFR | Amplification common in primary GBM |
| PTEN | Loss associated with aggressive behavior |
| TP53 | Mutations in ~28% of GBM cases |
Treatment Options for GBM
GBM treatment requires a multimodal approach combining surgery, radiation, and chemotherapy. Additionally, emerging therapies including immunotherapy offer promising new avenues for treatment.
Surgery
Maximal safe resection forms the cornerstone of GBM treatment. Neurosurgeons aim to remove as much tumor as possible while preserving neurological function. Greater extent of resection correlates with improved survival.
Learn More →Radiation Therapy
Following surgery, patients typically receive 60 Gy of radiation delivered over 6 weeks. Radiation targets remaining tumor cells while minimizing damage to surrounding brain tissue.
Learn More →Chemotherapy
Temozolomide (TMZ) is the standard chemotherapy agent, administered concurrently with radiation and continued as maintenance therapy. MGMT methylation status predicts response.
Learn More →Neurosurgical Resection of GBM
Surgery serves as the primary treatment for GBM, aiming to achieve maximal safe resection while preserving critical neurological functions. Research consistently demonstrates that greater extent of resection correlates with improved patient outcomes.
Goals of Surgery
- Remove maximum tumor volume safely
- Obtain tissue for diagnosis and molecular analysis
- Reduce mass effect and intracranial pressure
- Improve neurological symptoms
- Enhance effectiveness of subsequent treatments
Advanced Surgical Techniques
Modern GBM surgery utilizes sophisticated technologies including intraoperative MRI, neuronavigation, fluorescence-guided surgery (5-ALA), and awake craniotomy for tumors near eloquent areas. These advancements maximize resection while minimizing neurological deficits.
Microsurgical resection of glioblastoma demonstrating precision neurosurgical technique
Prof. Dr. Serdar Baki Albayrak
Internationally recognized for advanced glioma surgery and pioneering translational research in NK cell immunotherapy.

Neurosurgical Excellence
Prof. Dr. Serdar Baki Albayrak brings extensive experience in complex glioma surgery, including resections in eloquent cortex, deep-seated lesions, and brainstem tumors. His surgical philosophy prioritizes maximal safe resection combined with advanced microsurgical techniques.
Areas of Expertise
- Complex brain tumor surgery in eloquent areas
- Awake craniotomy for tumors near speech/motor areas
- Intraoperative neurophysiological monitoring
- Fluorescence-guided tumor resection
- NK cell immunotherapy research
Immunotherapy for Glioblastoma
Immunotherapy represents one of the most promising frontiers in GBM treatment. By harnessing the body's immune system, researchers aim to overcome the immunosuppressive tumor microenvironment characteristic of GBM.
The Immunotherapy Revolution
GBM creates a profoundly immunosuppressive microenvironment that allows tumor cells to evade immune detection. Overcoming this immunosuppression represents a major focus of modern cancer research.
Various immunotherapeutic strategies are under investigation, including checkpoint inhibitors, vaccine therapies, CAR-T cells, and Natural Killer (NK) cell therapy. Each approach offers unique advantages in the fight against GBM.
Why Immunotherapy for Brain Tumors?
- Potential to target infiltrating tumor cells surgery cannot reach
- May provide long-lasting immune memory against recurrence
- Can be personalized to individual tumor characteristics
- Generally better tolerated than conventional chemotherapy
NK Cell Therapy
Natural Killer (NK) cells are innate immune lymphocytes capable of recognizing and destroying tumor cells without prior sensitization. They play a crucial role in tumor immunosurveillance.
Tumor-Immunized Autologous NK Cell Therapy
This innovative approach involves isolating NK cells from the patient, exposing them to tumor antigens in the laboratory, and re-administering them to enhance tumor-specific cytotoxicity.
Prof. Dr. Albayrak's pioneering research has demonstrated the feasibility and safety of this personalized immunotherapy approach in high-grade glioma patients.
Published Research Findings
Clinical application has demonstrated: feasibility of tumor-specific NK cell activation, safety of intravenous autologous NK cell infusion, and successful integration of neurosurgery with immunotherapy.
Ongoing Clinical Trials
Multiple clinical trials are currently underway worldwide to evaluate the efficacy of NK cell-based immunotherapies in glioblastoma patients. These trials investigate various approaches including allogeneic NK cells, CAR-NK cells engineered to target GBM-specific antigens, and combination protocols integrating NK cell therapy with standard treatments. Promising early-phase results continue to drive expansion of clinical research in this field. Learn more about NK Cell Therapy →
Frequently Asked Questions
Comprehensive answers to the most common questions about glioblastoma multiforme, diagnosis, treatment, and prognosis.
The median survival for GBM patients receiving standard treatment (surgery, radiation, and temozolomide) is approximately 14-15 months. However, individual outcomes vary significantly based on age, tumor location, extent of resection, molecular markers (especially MGMT status), and overall health. Some patients survive several years, particularly those with favorable prognostic factors.
Currently, GBM cannot be completely cured due to its highly infiltrative nature. Even after successful surgery, microscopic tumor cells remain in surrounding brain tissue, leading to recurrence in 75-90% of cases. However, ongoing research in immunotherapy, targeted therapy, and personalized medicine offers hope for improved outcomes.
The exact cause of GBM remains largely unknown. The only confirmed risk factor is prior exposure to high-dose ionizing radiation. Certain rare genetic syndromes (Li-Fraumeni, neurofibromatosis) increase risk. Research has NOT established links between GBM and cell phones, electromagnetic fields, head injuries, or dietary factors.
Common early symptoms include persistent headaches (especially morning headaches), new-onset seizures, progressive weakness on one side of the body, speech difficulties, vision changes, memory problems, and personality changes. Symptoms depend on tumor location and may develop gradually or appear suddenly.
Most GBM cases (90-95%) are not hereditary and occur sporadically. However, certain inherited genetic syndromes increase GBM risk, including Li-Fraumeni syndrome, neurofibromatosis type 1, Lynch syndrome, and tuberous sclerosis. If you have a family history of brain tumors, genetic counseling may be beneficial.
The Stupp protocol is the current standard of care for newly diagnosed GBM, established in 2005. It consists of: maximal safe surgical resection, followed by 6 weeks of concurrent radiation therapy (60 Gy) plus daily temozolomide, followed by 6-12 cycles of maintenance temozolomide (5 days every 28 days). This regimen significantly improved survival compared to radiation alone.
MGMT (O6-methylguanine-DNA methyltransferase) is a DNA repair enzyme. When the MGMT gene promoter is methylated (silenced), the enzyme is not produced, making tumor cells more susceptible to temozolomide chemotherapy. Approximately 40-50% of GBM patients have MGMT promoter methylation, and these patients typically have better treatment response and longer survival.
Yes, numerous clinical trials are ongoing for GBM patients, testing new therapies including immunotherapy, targeted drugs, and novel treatment combinations. Clinical trials may be available for newly diagnosed patients, recurrent GBM, or specific molecular subtypes. Discuss trial options with your neuro-oncology team.
Expert Care for Glioblastoma Patients
Glioblastoma requires the highest level of neurosurgical expertise combined with innovative translational research. Our team offers comprehensive care integrating advanced surgical techniques with cutting-edge immunotherapy approaches.
Schedule a Consultation →
