NK Cell Therapy for Glioblastoma
(Ongoing Research)
Natural Killer cell immunotherapy represents one of the most promising frontiers in glioblastoma treatment today. In particular, this guide explores how NK cells recognize and destroy brain tumor cells. Additionally, it reveals what the latest clinical evidence shows. Furthermore, it explains why autologous expanded NK cell therapy may change the treatment landscape for GBM patients.
Moreover, patients and families deserve clear, science-based information about emerging treatment options. Therefore, this resource covers everything from the fundamental biology of NK cells to ongoing clinical trials. As a result, you will gain a comprehensive understanding of real-world outcomes and available options.
NK cells actively targeting glioblastoma cells through immunotherapy
What Are Natural Killer Cells?
Natural Killer cells serve as one of the immune system's most powerful first-line defenders against cancer and viral infections. Moreover, understanding their biology reveals why they hold such promise for brain tumor treatment.
The Body's Innate Cancer Fighters
Natural Killer (NK) cells belong to a group of immune cells known as innate lymphoid cells. Essentially, they originate from multipotent hematopoietic stem cells in the bone marrow. As a result, they mature into highly specialized tumor-surveillance agents. Unlike T cells and B cells, NK cells do not require prior exposure to a specific antigen. Consequently, they can respond immediately to cancer cells without delays.
Where NK Cells Are Found
In healthy individuals, NK cells constitute approximately 5 to 15 percent of all peripheral blood lymphocytes. Moreover, they circulate through the bloodstream and also reside in the spleen, liver, and lungs. Scientists classify them into two major subtypes. Specifically, CD56dim CD16+ cells exhibit potent cytotoxic activity. On the other hand, CD56bright CD16- cells primarily produce cytokines that coordinate broader immune responses.
How NK Cells Detect Cancer
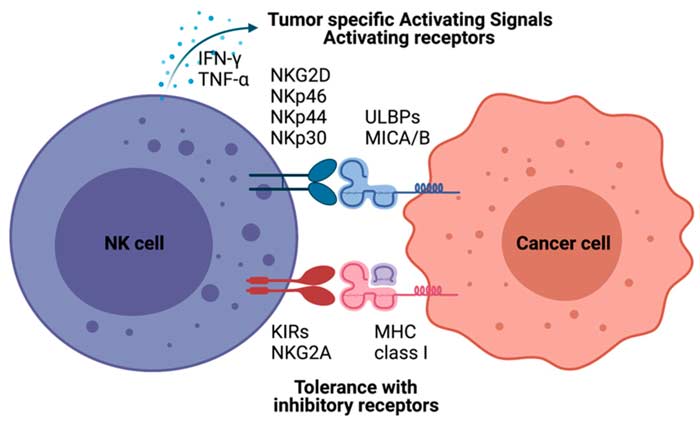
What makes NK cells particularly valuable in cancer therapy is their ability to distinguish healthy cells from abnormal ones. In essence, they achieve this through a sophisticated balance of activating and inhibitory receptor signals. For instance, when a cell displays markers of stress or malignant transformation, the activating signals override the inhibitory ones. Therefore, the NK cell proceeds to destroy the target.
Key Insight
Importantly, NK cells can eliminate cancer stem cells, including highly malignant glioblastoma stem cells. Notably, conventional therapies often fail to target these cells. As a result, this ability positions NK cell therapy as a crucial strategy for preventing tumor recurrence.
NK Cell Profile
| Origin | Bone Marrow Stem Cells |
| Surface Markers | CD56+, CD3- |
| Blood Percentage | 5-15% of Lymphocytes |
| Activation Speed | Immediate (No Priming Needed) |
| MHC Requirement | Not Required |
| Key Cytokines | IFN-gamma, TNF, GM-CSF |
NK Cell Subtypes
CD56dim CD16+ (Cytotoxic)
Essentially, these cells represent the majority of blood NK cells. Moreover, they deliver powerful cytotoxic responses and mediate antibody-dependent cellular cytotoxicity (ADCC). Consequently, they serve as the primary effectors in cancer cell elimination.
CD56bright CD16- (Regulatory)
In contrast, these cells predominate in tissues and lymph nodes. Furthermore, they produce high levels of cytokines that orchestrate broader immune responses. In addition, they can acquire cytotoxic functions upon appropriate stimulation.
How NK Cells Fight Cancer
NK cells employ multiple sophisticated mechanisms to identify and eliminate tumor cells. In particular, understanding these pathways helps explain why NK cell therapy holds such potential for glioblastoma treatment.
Perforin-Granzyme Pathway
Essentially, NK cells release perforin, a protein that creates pores in the cancer cell membrane. Then, through these pores, granzyme enzymes enter and trigger programmed cell death (apoptosis). As a result, this mechanism destroys tumor cells from the inside out without harming surrounding healthy tissue.
Death Receptor Signaling
Notably, NK cells express FAS ligand (FASL) and TRAIL on their surface. When these molecules bind to corresponding receptors on cancer cells, they activate a cascade of internal signals. Consequently, this leads to tumor cell apoptosis. Moreover, this pathway operates independently of the perforin-granzyme system.
ADCC (Antibody-Dependent Cytotoxicity)
Through CD16 receptors, NK cells recognize antibodies coating cancer cells. Then, they launch a targeted attack. This antibody-dependent cellular cytotoxicity mechanism bridges innate and adaptive immunity. As a result, it amplifies the overall antitumor response significantly.
Receptor Balance: The Missing Self Hypothesis
Notably, NK cells use a unique surveillance system to detect cancer cells. Every healthy cell displays MHC class I molecules on its surface. Essentially, these serve as a "self" identification. In addition, NK cells carry inhibitory receptors (such as KIR and NKG2A) that recognize these MHC molecules. Therefore, when they encounter a healthy cell, the inhibitory signals prevent an attack.
Why Cancer Cells Become Vulnerable
However, many cancer cells downregulate or lose their MHC class I expression. Essentially, they do this to evade T cell detection. Ironically, however, this evasion strategy makes them vulnerable to NK cells. Without the inhibitory signal, NK cell activating receptors (NKG2D, NKp30, NKp44, NKp46, and DNAM-1) take over. As a result, these receptors detect stress ligands that cancer cells express. Consequently, the balance shifts toward activation.
This "missing self" detection mechanism makes NK cells particularly effective against resistant tumors. Furthermore, it explains why NK cell therapy and T cell therapy can complement each other. In summary, this dual approach strengthens comprehensive treatment strategies.
Cytokine Production and Immune Coordination
Beyond direct killing, NK cells produce cytokines that amplify the immune response. IFN-gamma is one of the primary cytokines released by activated NK cells. Notably, it accomplishes several critical functions. For example, it enhances the antigen-presenting capability of dendritic cells. Moreover, it also promotes T helper cell activation. Additionally, it directly inhibits tumor cell proliferation and angiogenesis.
TNF-alpha, another key cytokine, triggers inflammation within the tumor microenvironment. As a result, this inflammation recruits additional immune cells. Meanwhile, GM-CSF stimulates the production of new immune cells from bone marrow precursors. In conclusion, these cytokines create a cascade of immune activation that extends far beyond the initial NK cell response.
Clinical Significance
Importantly, research demonstrates that higher NK cell infiltration in brain tumors correlates with improved patient outcomes. As a result, this finding supports the therapeutic strategy of boosting NK cell numbers and activity through adoptive cell therapy.
NK Cells in the Glioblastoma Microenvironment
Glioblastoma creates one of the most immunosuppressive tumor environments in the body. Therefore, understanding how this affects NK cells is essential for developing effective immunotherapy strategies.
The Immunosuppressive Challenge
Glioblastoma tumors actively suppress the immune system through multiple mechanisms. For instance, tumor-associated macrophages (TAMs) account for up to 30 percent of the cellular population within GBM. As a result, they release immunosuppressive cytokines that dampen NK cell activity. In addition, regulatory T cells and myeloid-derived suppressor cells further weaken the antitumor immune response.
TGF-beta and IDO Pathways
The glioblastoma microenvironment also produces TGF-beta, a potent immunosuppressive factor. This directly reduces NK cell cytotoxicity. Moreover, GBM cells convert tryptophan to kynurenine through the IDO enzyme. Consequently, this inhibits NK cell function while promoting regulatory T cell recruitment. Together, these mechanisms make glioblastoma particularly challenging for immunotherapy.
Treatment-Induced Immune Suppression
Standard treatments also contribute to immune suppression. Specifically, temozolomide chemotherapy, radiation therapy, and corticosteroids all reduce lymphocyte counts. Therefore, treatment-induced lymphopenia creates an additional barrier. In other words, there is less effective immune surveillance against residual or recurring tumor cells.
Why NK Cells Still Hold Promise
Despite these challenges, NK cells offer unique advantages. Unlike T cells, they do not require MHC-mediated antigen presentation. Furthermore, NK cells can target glioblastoma stem cells, the treatment-resistant cells responsible for tumor recurrence. In particular, research shows that GBM stem cells express stress ligands. As a result, they become susceptible to NK cell-mediated killing.
Additionally, surgical resection reduces the overall immunosuppressive burden. For example, studies show increased NKG2D expression in post-surgical samples. This finding suggests that removing the primary tumor partially restores NK cell function. Therefore, it creates a window of opportunity for NK cell therapy.
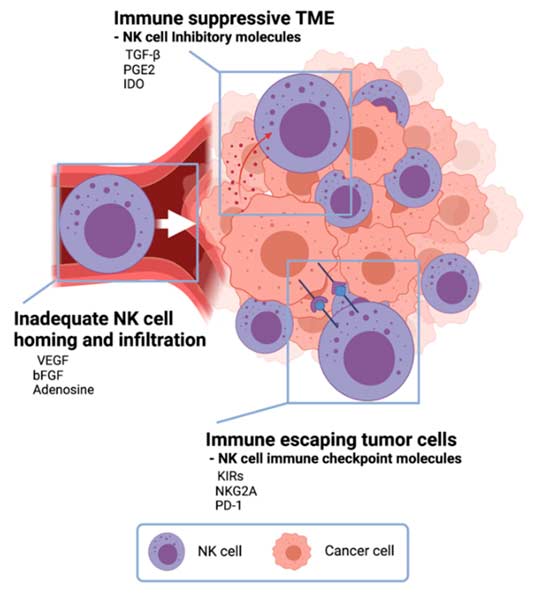
Illustration of NK cells navigating the glioblastoma tumor microenvironment
Tumor Immune Evasion Mechanisms
- In particular, TGF-beta secretion that directly suppresses NK cell cytotoxicity
- Additionally, MHC class I upregulation to engage NK inhibitory receptors
- Furthermore, shedding of NKG2D ligands to reduce activating signals
- Moreover, IDO-mediated tryptophan depletion and kynurenine production
- Similarly, PD-L1 expression on tumor cells and associated macrophages
- Also, recruitment of immunosuppressive regulatory T cells
- Finally, extracellular matrix barriers limiting immune cell infiltration
Autologous Expanded NK Cell Therapy
Tumor-immunized autologous NK cell therapy represents a personalized approach. Specifically, it uses the patient's own immune cells to fight glioblastoma. Moreover, this section explains the science behind the expansion process and its clinical applications.
The Expansion Process
Autologous NK cell therapy begins with collecting blood from the patient. Then, laboratory technicians isolate NK cells from peripheral blood mononuclear cells using immunomagnetic selection. Once isolated, these cells enter a carefully controlled expansion protocol. As a result, this process multiplies their numbers while enhancing antitumor activity.
Cytokine Stimulation
During expansion, scientists stimulate NK cells with specific cytokines. These include IL-2, IL-15, and IL-21. Each cytokine serves a distinct purpose. For instance, IL-2 drives proliferation and enhances cytotoxic function. Meanwhile, IL-15 promotes NK cell survival and memory formation. Additionally, IL-21 amplifies antitumor effector functions.
Tumor-Immunized Approach
In the tumor-immunized approach, expanded NK cells receive additional exposure to the patient's own tumor antigens. Consequently, this exposure primes the NK cells to recognize specific molecular signatures. As a result, the re-infused cells demonstrate enhanced recognition and killing of the patient's glioblastoma cells.
The entire expansion process typically takes two to three weeks. During this time, NK cell numbers can increase by several hundred-fold. Before re-infusion, each batch undergoes rigorous quality control testing. Therefore, this meticulous process ensures patients receive a safe and effective therapeutic product.
Sources of NK Cells for Therapy
Autologous (Patient's Own)
These cells come from the patient's own blood. Therefore, they carry no risk of graft-versus-host disease. While they may show reduced function due to tumor-induced suppression, the expansion process restores and enhances their activity. In addition, the personalized approach ensures optimal compatibility.
Allogeneic (Donor-Derived)
Healthy donor NK cells often show greater baseline cytotoxicity. This is because they have not been exposed to tumor-induced suppression. However, they carry a theoretical risk of immune rejection. On the other hand, the HLA-mismatch between donor and recipient can actually enhance antitumor activity through the "missing self" mechanism.
Umbilical Cord Blood
Cord blood provides a readily available, cryopreservable source of NK cells. Although these cells are functionally immature, they can be expanded and activated. As a result, they achieve therapeutic potency. Moreover, the ease of collection makes them suitable for off-the-shelf products.
NK-92 Cell Line
This standardized cell line offers consistent quality and unlimited availability. In addition, NK-92 cells serve as a platform for genetic engineering, including CAR-NK development. However, they require irradiation before infusion to prevent uncontrolled proliferation.
Clinical Evidence for NK Cell Therapy (Ongoing Research)
Recent clinical studies demonstrate encouraging outcomes for NK cell-based therapies in glioblastoma. Although many trials remain in early phases, the results provide a strong foundation for continued development.
Landmark Pilot Study Results
In 2025, a pivotal pilot study evaluated a combination of ANKTIVA (an IL-15 agonist), NK cell therapy, and Tumor Treating Fields in five patients with recurrent glioblastoma. Notably, all five patients had previously failed standard-of-care treatment.
The results were remarkable. Specifically, all five patients achieved disease control. Moreover, three out of five patients responded to treatment. In particular, two achieved near-complete response. Furthermore, ANKTIVA treatment increased the absolute lymphocyte count in all five patients. Consequently, this effectively reversed the lymphopenia caused by prior radiation and chemotherapy.
The principal investigator stated that they had never experienced such near-complete responses in years of treating glioblastoma patients. As a result, these findings prompted the initiation of a randomized Phase 2 trial targeting second-line GBM patients.
Preclinical Foundations
Extensive preclinical research supports the clinical development of NK cell therapy. For instance, laboratory studies consistently demonstrate that activated NK cells can kill GBM cells. In addition, they can destroy treatment-resistant stem cells. Similarly, animal models show survival benefits when NK cell therapy combines with standard treatments.
Specifically, temozolomide increases the expression of NKG2D ligands on GBM cells through the DNA damage response pathway. Consequently, this upregulation makes tumor cells more recognizable. As a result, they become vulnerable to NK cell attack. Likewise, radiation therapy enhances NK cell-mediated killing by promoting ligand expression on surviving cancer cells.
Active Clinical Trials
Currently, multiple clinical trials evaluate NK cell therapy for brain tumors, including studies with autologous NK cells, allogeneic NK cells, CAR-NK cells targeting HER2 and EGFRvIII, and NK cell combinations with checkpoint inhibitors. Therefore, interested patients should visit ClinicalTrials.gov or consult their neuro-oncology team.
Disease Control
Notably, patients achieving disease control in pilot study
Near-Complete Response
Specifically, patients achieving near-complete tumor response
ALC Recovery
Moreover, patients with lymphocyte count restoration
Severe Toxicity
In addition, chemotherapy-free immune-stimulating approach
Combining NK Cells with Other Treatments
NK cell therapy shows the greatest potential when combined with other treatment modalities. In particular, research reveals powerful synergies that enhance overall antitumor efficacy.
Synergy with Standard Treatments
Surgical resection of glioblastoma reduces the tumor's immunosuppressive burden. As a result, it creates a favorable environment for NK cell activity. Moreover, studies show that post-surgical patients exhibit higher NKG2D receptor expression. This is primarily due to decreased tumor-derived TGF-beta levels. Therefore, the period following surgery represents an optimal window for NK cell therapy.
Temozolomide Synergy
Temozolomide chemotherapy also enhances NK cell effectiveness. Specifically, it causes DNA damage in tumor cells. Consequently, this triggers the DNA damage response pathway. As a result, the expression of NKG2D ligands increases on the cancer cell surface.
These ligands serve as "eat me" signals for NK cells. Furthermore, preclinical models demonstrate that combining temozolomide pretreatment with NK cell therapy achieves greater tumor control than either treatment alone.
Radiation Sensitization
Similarly, radiation therapy sensitizes residual GBM cells to NK cell-mediated killing. Although radiation reduces the total number of NK cells, it increases the functional capacity of surviving cells. In particular, their NKG2D-dependent IFN-gamma production improves. In conclusion, the net effect favors enhanced tumor recognition.
Timing Matters
The sequencing and timing of NK cell therapy critically affects outcomes. For instance, administering NK cells after chemotherapy-induced lymphopenia provides a homeostatic advantage. Therefore, careful treatment coordination by the medical team ensures optimal benefit.
Novel Combination Strategies
Checkpoint Inhibitors + NK Cells
Anti-PD-1 and anti-PD-L1 antibodies can relieve immunosuppressive braking signals within the tumor. When combined with NK cell infusion, checkpoint inhibitors allow the transferred cells to maintain their cytotoxic function. As a result, early clinical data supports this combination approach.
Oncolytic Virotherapy + NK Cells
Oncolytic viruses selectively infect and destroy tumor cells while sparing healthy tissue. In a triple-treatment approach, oncolytic herpes simplex virus combined with proteasome inhibitors and NK cells shows synergistic effects. Moreover, the virus causes immunogenic cell death. Consequently, this recruits and activates NK cells at the tumor site.
Tumor Treating Fields + NK Cells
Tumor Treating Fields (TTFields) deliver alternating electric fields that disrupt cancer cell division. Furthermore, recent pilot studies combining TTFields with NK cell therapy in recurrent GBM demonstrate disease control in all treated patients. In addition, the Fields may also enhance immune cell infiltration.
IL-15 Agonists + NK Cells
IL-15 superagonist complexes like ANKTIVA stimulate NK cell proliferation. Additionally, they enhance antitumor activity in vivo. As a result, clinical evidence shows that IL-15 agonist treatment reverses treatment-induced lymphopenia. Therefore, it rescues NK cell counts and function in glioblastoma patients.
CAR-NK Cell Technology (Ongoing Research)
Chimeric Antigen Receptor NK (CAR-NK) cells combine the innate killing power of NK cells with engineered precision targeting. Therefore, this technology addresses several limitations of both traditional NK cell therapy and CAR-T cell approaches.
Advantages Over CAR-T Cells
CAR-NK cells offer several critical advantages over CAR-T cell therapy. First, they carry a significantly lower risk of graft-versus-host disease (GVHD). As a result, this allows the use of allogeneic (donor-derived) cells. Consequently, it eliminates the need for patient-specific manufacturing. Moreover, it enables the creation of "off-the-shelf" products available immediately.
Safety Advantages
Second, CAR-NK cells demonstrate a much lower incidence of cytokine release syndrome (CRS) and neurotoxicity. Notably, for brain tumor patients, the reduced neurotoxicity risk is especially important. Third, NK cells retain their intrinsic cytotoxic mechanisms alongside the CAR-directed killing. Therefore, this provides a dual attack on tumor cells. In other words, even if the tumor downregulates the CAR-targeted antigen, NK cells can still destroy cancer cells through their native receptors.
Clinical Development
Currently, Phase I and Phase I/II trials evaluate CAR-NK cells targeting HER2 (NCT03383978) and MUC1 (NCT02839954) in glioma patients. Moreover, preclinical data also supports anti-EGFRvIII CAR-NK cells combined with CXCR4 expression for enhanced tumor homing.
CAR-NK cells engineered to target specific glioblastoma antigens
Key GBM Targets for CAR-NK
| EGFRvIII | Mutant receptor found in 25-30% of GBM |
| HER2 | Overexpressed in a subset of glioblastoma |
| MUC1 | Aberrantly expressed in solid tumors including GBM |
| GD2 | Ganglioside expressed on GBM stem cells |
Overcoming Treatment Challenges (NK Immunotherapy Ongoing Research)
Researchers actively develop solutions to the obstacles that limit NK cell therapy effectiveness in glioblastoma. Importantly, each challenge drives innovation that brings this treatment closer to clinical reality.
Blood-Brain Barrier Penetration
GBM disrupts the blood-brain barrier (BBB) locally. As a result, this allows some NK cell infiltration. Furthermore, researchers enhance delivery through intratumoral injection during surgery. In addition, they use focused ultrasound and chemokine receptors like CXCR4 for improved brain homing.
Tumor Immune Evasion
Scientists counter tumor evasion by combining NK cells with checkpoint inhibitors. Moreover, they engineer cells resistant to TGF-beta suppression. Additionally, oncolytic viruses create an inflammatory microenvironment. As a result, these strategies restore NK cell function within the hostile tumor milieu.
Persistence and Expansion In Vivo
To ensure NK cells survive long enough, researchers use cytokine support with IL-15 agonists. In addition, they create memory-like NK cells through brief cytokine preactivation. Furthermore, they optimize the timing of infusion relative to lymphodepleting regimens. Consequently, these approaches improve long-term NK cell persistence.
The NK Cell Treatment Process
Understanding the step-by-step treatment process helps patients and families prepare for NK cell immunotherapy. In addition, each phase involves specialized procedures designed to maximize therapeutic benefit.
First, the medical team assesses your overall health status. Then, they review imaging studies and determine candidacy for NK cell therapy. Additionally, they evaluate prior treatments and molecular characteristics of the tumor. As a result, they design a personalized treatment plan.
Next, a specialized blood draw collects peripheral blood mononuclear cells containing NK cells. Typically, this outpatient procedure takes approximately three to four hours. Afterward, the collected cells are transported to the processing laboratory under controlled conditions.
Subsequently, laboratory technicians separate NK cells from other blood components using immunomagnetic selection. Then, the isolated cells then enter the expansion protocol with cytokine stimulation. Moreover, during this phase, cells may also receive exposure to tumor antigens for immunization.
During this critical phase, NK cells multiply several hundred-fold under carefully controlled conditions. In addition, quality assessments monitor cell viability, purity, and cytotoxic function. As a result, this step ensures adequate numbers of highly active NK cells for infusion.
Depending on the protocol, patients may receive a lymphodepleting regimen. Essentially, this creates space for the infused NK cells. Furthermore, it reduces immunosuppressive cell populations. Typically, the conditioning phase occurs one to three days before NK cell infusion.
Finally, expanded NK cells are infused intravenously or delivered directly to the tumor site. Subsequently, post-infusion monitoring tracks vital signs and immune cell counts. Generally, most patients tolerate the infusion well. In general, the most common side effects include mild fever and temporary fatigue.
Prof. Dr. Serdar Baki Albayrak
As a leading neurosurgeon and researcher, he specializes in glioblastoma treatment with pioneering work in NK cell immunotherapy for malignant brain tumors.

Pioneering NK Cell Research for Brain Tumors
Prof. Dr. Serdar Baki Albayrak brings decades of neurosurgical expertise to the field of glioblastoma treatment. In particular, his research on expanded autologous NK cell therapy for malignant gliomas represents a significant contribution to translational neuro-oncology. Moreover, by integrating advanced surgical techniques with innovative immunotherapy approaches, he provides patients with comprehensive, multimodal care.
Specifically, his work focuses on optimizing NK cell expansion protocols for brain tumor patients. Additionally, he develops tumor-immunized NK cell products. Furthermore, he identifies the optimal treatment timing and sequencing. As a result, this research contributes to the growing body of evidence supporting NK cell therapy as a viable complement to standard GBM treatment.
As a clinician-scientist, he bridges the gap between laboratory research and patient care. Therefore, he ensures that scientific advances translate into practical treatment options. In addition, his approach considers each patient's unique tumor biology and treatment history when designing individualized immunotherapy strategies.
FAQ — NK Cell Therapy for Glioblastoma (Ongoing Research)
Patients and healthcare professionals frequently ask these questions about NK cell immunotherapy. Below you will find evidence-based answers to help you make informed decisions.
General Questions About NK Cell Therapy
NK cell therapy uses Natural Killer cells to find and destroy glioblastoma tumor cells. First, the medical team collects NK cells from the patient or a donor. Then, scientists expand and activate them in the laboratory. As a result, they create a concentrated army of cancer-fighting cells ready for infusion. Moreover, the expanded cells recognize glioblastoma through stress ligands and missing MHC signals. Finally, they destroy tumor cells through perforin-granzyme release and death receptor signaling. Consequently, this approach works independently of the mechanisms that tumor cells use to evade T cell detection.
NK cell therapy generally carries a favorable safety profile. Common side effects include mild fever, temporary fatigue, and brief flu-like symptoms. These usually resolve within a few days. In comparison to CAR-T cell therapy, NK cell treatment poses significantly lower risks of cytokine release syndrome (CRS), neurotoxicity, and graft-versus-host disease. Furthermore, serious adverse events remain rare in clinical trials reported to date. Therefore, each patient's medical team monitors closely and manages any side effects promptly.
Expanded autologous NK cells undergo a specialized process. This process enhances both their quantity and quality. Initially, scientists isolate NK cells from the patient's own blood. Next, they stimulate them with cytokines (IL-2, IL-15, IL-21). As a result, the cells multiply several hundred-fold over two to three weeks. In the tumor-immunized variant, the cells also receive exposure to the patient's specific tumor antigens. Consequently, this primes them for more effective targeting. In contrast, standard NK cell therapy may use unmodified or minimally expanded cells.
Yes, and in fact combination approaches show the most promise. First, surgery reduces tumor burden and relieves immunosuppression. As a result, it creates a better environment for NK cell activity. Additionally, temozolomide chemotherapy upregulates stress ligands on tumor cells through the DNA damage response. Consequently, this makes them more recognizable to NK cells. Similarly, radiation enhances tumor visibility to NK cells. However, the timing and sequencing of these treatments must be carefully coordinated to maximize synergistic benefits.
Clinical Evidence and Trial Information
Active clinical trials can be found on ClinicalTrials.gov. You can search for "NK cells glioblastoma" or "natural killer cells brain tumor." Moreover, major cancer centers conduct these studies, including MD Anderson, Memorial Sloan Kettering, and various university hospitals in the US and Europe. Current trials evaluate autologous NK cells, allogeneic NK cells, and CAR-NK cells targeting HER2 and EGFRvIII. In addition, your neuro-oncology team can help identify trials matching your situation.
While large-scale randomized trial data is still emerging, early-phase results show encouraging signs. For instance, a recent pilot study demonstrated 100 percent disease control in five recurrent GBM patients. These patients received ANKTIVA (IL-15 agonist), NK cell therapy, and Tumor Treating Fields. Notably, two patients achieved near-complete response. As a result, these preliminary findings have prompted a larger Phase 2 randomized trial. However, it is important to note that individual results vary based on tumor characteristics and overall patient health.
The cost varies based on the treatment protocol, NK cell source, and treatment location. For example, autologous NK cell expansion requires specialized GMP-grade laboratory facilities. Consequently, this contributes to higher costs. On the other hand, clinical trial participation may provide treatment at no direct cost. Furthermore, as off-the-shelf NK cell products advance through development, production economies of scale are expected to reduce costs. Therefore, patients should discuss financial planning with their treatment center.
The blood-brain barrier presents a physical challenge for NK cell delivery. However, glioblastoma disrupts the barrier locally. As a result, this allows some degree of immune cell infiltration. In addition, researchers employ several strategies to enhance delivery. These include intratumoral injection during surgical resection, focused ultrasound, and genetic engineering of NK cells. Specifically, expressing chemokine receptors like CXCR4 promotes migration toward brain tumors. Currently, these approaches are under active clinical investigation.
Access, Cost, and Research Landscape
Several organizations drive NK cell therapy development. ImmunityBio leads with their ANKTIVA platform and PD-L1 t-haNK product. In addition, Fate Therapeutics develops iPSC-derived NK cell products. Meanwhile, Nkarta focuses on engineered NK cell therapies with enhanced tumor targeting. Furthermore, academic institutions including Johann W. Goethe University Hospital conduct CAR-NK trials. As a result, these companies and institutions represent a growing field dedicated to making NK cell therapy accessible worldwide.
Current leading approaches include autologous expanded NK cells activated with cytokine cocktails. Additionally, allogeneic NK cells from healthy donors or cord blood show promise. Moreover, the ANKTIVA plus NK cell combination protocol demonstrates strong results. CAR-NK cells targeting specific antigens also represent a major advance. Finally, cytokine-induced memory-like NK cells offer enhanced persistence. For glioblastoma specifically, combination approaches that include surgical resection show the most promising clinical outcomes.
Expert NK Cell Therapy for Glioblastoma — Ongoing Research
NK cell immunotherapy represents a transformative approach to glioblastoma treatment. Moreover, our team combines advanced neurosurgical expertise with cutting-edge immunotherapy research. As a result, we offer patients comprehensive, personalized care. Therefore, contact us to learn whether NK cell therapy may be appropriate for your situation.
Schedule a Consultation →
